Nuclear Membrane Repairs the ‘Dark Matter’ of DNA
Once thought of simply as a barrier to keep DNA inside the nucleus of a cell, the membrane actually helps to fix catastrophically broken DNA strands
Contact: Robert Perkins at (213) 740-9226 or perkinsr@usc.edu
Scientists have found a new function of the nuclear membrane, the envelope that encases and protects DNA in the nucleus of a cell — it fixes potentially fatal breaks in DNA strands.
Previously, the nuclear membrane was thought to be mostly just a protective bubble around the nuclear material, with pores acting as channels to transport molecules in and out. But in a study published on October 26 in Nature Cell Biology, a research team led by Irene Chiolo documents how broken strands of a portion of DNA known as heterochromatin are dragged to the nuclear membrane for repair.
DNA exists inside of a cell’s nucleus in two forms: euchromatin and heterochromatin. Euchromatin gets all of the attention because it encodes most of the genome, while heterochromatin, which is mostly composed of repeated DNA sequences, has long been ignored as “junk DNA.”
“Scientists are now starting to pay a lot of attention to this mysterious component of the genome,” said Chiolo, assistant professor at the USC Dornsife College of Letters, Arts and Sciences. “Heterochromatin is not only essential for chromosome maintenance during cell division; it also poses specific threats to genome stability. Heterochromatin is potentially one of the most powerful driving forces for cancer formation, but it is the ‘dark matter’ of the genome. We are just beginning to unravel how repair works here.”
The reason why we don’t experience thousands of cancers every day in our body is because we have incredibly efficient molecular mechanisms that repair the frequent damages occurring in our DNA. But those that work in heterochromatin are quite extraordinary.
“Repeated sequences tend to recombine with each other during DNA repair,” said Taehyun Ryu, USC graduate student and first author on this study. “This would lead to chromosome aberrations as frequently observed in cancer cells. What prevents these outcomes in normal cells was unclear.”
“We knew that nuclear membrane dysfunctions are common in cancer cells,” Chiolo said. “Our studies now suggest how these dysfunctions can affect heterochromatin repair and have a causative role in cancer progression.”
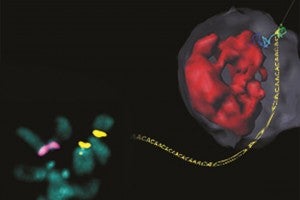
Working with the fruit fly Drosophila melanogaster, the team observed that breaks in heterochromatin are repaired after damaged sequences move away from the rest of the chromosome to the inner wall of the nuclear membrane. There, a trio of proteins mends the break in a safe environment, where it cannot accidentally get tangled up with incorrect chromosomes.
This study may help reveal how and why organisms become more predisposed to cancer as they age — the nuclear membrane progressively deteriorates as an organism ages, removing this bulwark against genome instability.
Next, Chiolo, Ryu and their colleagues will explore how the movement of broken sequences is accomplished and regulated, and what happens in cells and organisms when this membrane-based repair mechanism fails. Their ultimate goal is to understand how this mechanism functions in human cells and identify new strategies to prevent their catastrophic failure and cancer formation.
Chiolo and Ryu collaborated with USC’s Brett Spatola, Laetitia Delabaere, Kate Bowlin and Hannah Hopp, and Lawrence Berkely National Lab’s Gary Karpen and Ryan Kunitake. This research was funded by the USC Gold Family Fellowship, the USC Research Enhancement Fellowship, the USC Provost Fellowship, the Rose Hills Foundation, and grants from the National Institute of Health (NIH R21ES021541, R01GM117376 and R01GM08661).
The paper can be found online here: http://www.nature.com/ncb/journal/vaop/ncurrent/full/ncb3258.html
Image: Composite illustration shows the location of heterochromatin in a chromosome (highlighted in pink and yellow, bottom left) and in a nucleus (red, top right). The blue-to-green line in the upper right-hand corner shows the path that a damaged section takes as it is dragged to the nuclear membrane (gray) for repair. (Courtesy of Irene Chiolo)
###